Sodium metavanadate
 | |
| Names | |
|---|---|
| IUPAC name
Sodium trioxovanadate(V)
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.033.869 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3285 (SODIUM VANADATE) |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
NaVO3 |
| Molar mass | 121.9295 g/mol |
| Appearance | yellow crystalline solid |
| Density | 2.84g/cm3 |
| Melting point | 630 °C (1,166 °F; 903 K) |
Solubility in water
|
19.3 g/100 mL (20 °C) 40.8 g/100 mL (80 °C) |
| Thermochemistry | |
Heat capacity (C)
|
97.6 J/mol K |
Std molar
entropy (S⦵298) |
113.8 J/mol K |
Std enthalpy of
formation (ΔfH⦵298) |
−1148 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic, irritant |
| GHS labelling:[1] | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H301, H315, H319, H332, H335, H361, H361d, H372, H411 |
Precautionary statements
|
P203, P260, P264, P264+P265, P270, P271, P273, P280, P301+P316, P302+P352, P304+P340, P305+P351+P338, P317, P318, P319, P321, P330, P332+P317, P337+P317, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 
2
0
0 |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
98 mg/kg (rat, oral) |
| Related compounds | |
Other anions
|
|
Other cations
|
Ammonium metavanadate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
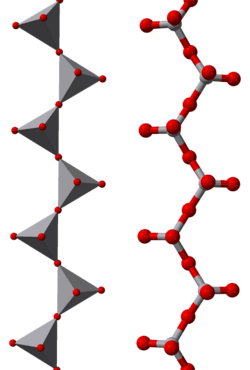
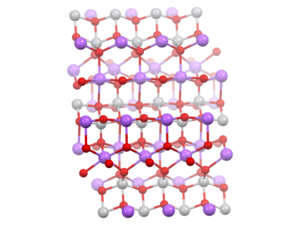
Sodium metavanadate is the inorganic compound with the formula NaVO3.[2] It is a yellow, water-soluble salt.
Sodium metavanadate is a common precursor to other vanadates. At low pH it converts to sodium decavanadate. It is also precursor to exotic metalates such as [γ-PV2W10O40]5-, [α-PVW11O40]4-, and [β-PV2W10O40]5-.[3]
Minerals
Sodium metavanadate occurs as two minor minerals, metamunirite (anhydrous) and a dihydrate, munirite. Both are very rare, metamunirite is now known only from vanadium- and uranium-bearing sandstone formations of central-western USA and munirite from Pakistan and South Africa.[4]
References
- ^ PubChem. "Sodium metavanadate". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-02-05.
- ^ Kato, K.; Takayama, E. (1984). "Das Entwässerungsverhalten des Natriummetavanadatdihydrats und die Kristallstruktur des beta-Natriummetavanadats" [The dehydration activity of sodium metavanadate dihydrate and the crystal structure of β-sodium metavanadate]. Acta Crystallogr. B40 (2): 102–105. Bibcode:1984AcCrB..40..102K. doi:10.1107/S0108768184001828.
- ^ Domaille, Peter J. (2007). "Vanadium(V) Substituted Dodecatungstophosphates". Inorganic Syntheses. Vol. 27. pp. 96–104. doi:10.1002/9780470132586.ch17. ISBN 9780470132586.
- ^ "Munirite". Mindat.